Acid and Base Theories
Arrhenius Theory:
The Arrhenius Theory was created in 1884 by Svante Arrhenius, which was the the first modern definition of acids and bases. His theory that substances stay ionized constantly is now something that is accepted without question, but during the late 1800s, this claim was considered bizarre. As a result, Arrhenius had to defend his theory for years and he was forced to explain his ideas numerous times to those who opposed the cutting edge break through. In the end, all of his hard work became beneficial when fellow scientists eventually did accept his claim earning him the nobel prize in Chemistry in 1903. His theory is now a basis of identifying acid and bases from their molecular structure. The theory describes a way that acids and bases can be distinguished from each other by their molecular structures. This theory was extremely important during the past because there was not a simple system of distinguishing acids and bases from each other but this breakthrough allowed for ease to ensue. Because of this theory, students and scientists have an easy time making sense of equations because they are immediately able to see what types of chemicals are reacting with each other.
Examples:
H3POH4 NH4OH KBr KOH H2O= HOH CH4
Acid Base Neutral Base Neutral Neutral
***In the case of H2O, the molecular structure is actually HOH, meaning that it is both an acid and a base. Because the molecule is both an acid and a base, it becomes positive as a result.
***In the case of CH4 the substance is neutral because the hydrogen is not positive, it is negative which is why it is written at the end of the compound. As a result, the molecule does not contain either a H+ ion or a OH- ion.
Brønsted-Lowry Theory:
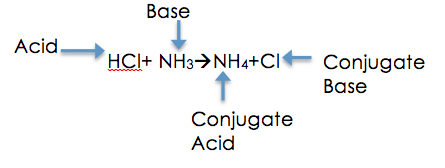
The Brønsted-Lowry Theory, also known as the Proton Theory of Acids and Bases, was created by Johannes Nicolaus Brønsted and Thomas Martin Lowry in 1923. The theory had been created separately between the two scientists when there was a lot of confusion surrounding the Arrhenius Theory, as many were confused how the acids and bases reacted and changed during a reaction between the two. As a result, both of the scientists decided to start investigations in attempts to uncover the movements of protons and how the acids and bases change during their reactions. The main concept of the theory is to describe the reaction between an acid and a base. The theory says that during an acid and base reaction there is a transfer of H+ ions. When the acid loses its proton during the reaction, it becomes a base on the product side, but not just any base a conjugate base. This also means that a base becomes an acid when it receives the H+ ion, thus becoming a conjugate acid. This theory is extremely important because it allows scientists to understand chemical reactions in a whole new light, the idea that acids and bases are dependent on each other for their reactions is key. The theory was crucial during the 1920s because there had been a lot of confusion about the movement of protons and how the acids and cases change during their reactions. This theory brought light into the confusion and allows us today to fully understand reactions between the acids and bases.
The Arrhenius Theory was created in 1884 by Svante Arrhenius, which was the the first modern definition of acids and bases. His theory that substances stay ionized constantly is now something that is accepted without question, but during the late 1800s, this claim was considered bizarre. As a result, Arrhenius had to defend his theory for years and he was forced to explain his ideas numerous times to those who opposed the cutting edge break through. In the end, all of his hard work became beneficial when fellow scientists eventually did accept his claim earning him the nobel prize in Chemistry in 1903. His theory is now a basis of identifying acid and bases from their molecular structure. The theory describes a way that acids and bases can be distinguished from each other by their molecular structures. This theory was extremely important during the past because there was not a simple system of distinguishing acids and bases from each other but this breakthrough allowed for ease to ensue. Because of this theory, students and scientists have an easy time making sense of equations because they are immediately able to see what types of chemicals are reacting with each other.
- All acid molecules dissociate in water and yield H+ ions, meaning that any compound that contains at least one H+ ion is an acid.
- Examples: HCl, HBr, H2SO4
- The hydrogen molecule always is placed first in the compound because it is the positive ion in the ionic bond, positive elements always go first.
- All base molecules dissociate in water and yield OH- ions, meaning that any compound that contains at least one OH- ion is an base.
- Examples: NaOH, LiOH, Mg(OH)2
- The OH molecule goes that the end of the compound because it is the negative part of the ionic bond
- The OH molecule is a polyatomic ion, which is an ion that has two atoms with a charge. Whenever there are parentheses in an equation, the compound that is inside of it will always be a polyatomic atom.
- Examples: NaOH, LiOH, Mg(OH)2
- Examples: HCl, HBr, H2SO4
- All neutral molecules either contain both H+ and OH- ions or the molecules contain neither of them.
Examples:
H3POH4 NH4OH KBr KOH H2O= HOH CH4
Acid Base Neutral Base Neutral Neutral
***In the case of H2O, the molecular structure is actually HOH, meaning that it is both an acid and a base. Because the molecule is both an acid and a base, it becomes positive as a result.
***In the case of CH4 the substance is neutral because the hydrogen is not positive, it is negative which is why it is written at the end of the compound. As a result, the molecule does not contain either a H+ ion or a OH- ion.
Brønsted-Lowry Theory:
The Brønsted-Lowry Theory, also known as the Proton Theory of Acids and Bases, was created by Johannes Nicolaus Brønsted and Thomas Martin Lowry in 1923. The theory had been created separately between the two scientists when there was a lot of confusion surrounding the Arrhenius Theory, as many were confused how the acids and bases reacted and changed during a reaction between the two. As a result, both of the scientists decided to start investigations in attempts to uncover the movements of protons and how the acids and bases change during their reactions. The main concept of the theory is to describe the reaction between an acid and a base. The theory says that during an acid and base reaction there is a transfer of H+ ions. When the acid loses its proton during the reaction, it becomes a base on the product side, but not just any base a conjugate base. This also means that a base becomes an acid when it receives the H+ ion, thus becoming a conjugate acid. This theory is extremely important because it allows scientists to understand chemical reactions in a whole new light, the idea that acids and bases are dependent on each other for their reactions is key. The theory was crucial during the 1920s because there had been a lot of confusion about the movement of protons and how the acids and cases change during their reactions. This theory brought light into the confusion and allows us today to fully understand reactions between the acids and bases.
- All acids are proton donors during a reaction, meaning that the acid gives away a H+ ion to the base.
- All bases are proton receivers during a reaction, meaning the base receives the H+ ion from the acid.
- Conjugate Acid= the base after it receives a H+ ion
- Conjugate Base= the acid after it gives away a H+ ion
Lewis Theory:
Once the Brønsted-Lowry Theory was created, which brought a description of the movement of protons in an acid base reaction, scientists wondered what happened to the electrons during the reaction. The Lewis Theory, created by Gilbert N. Lewis, is another theory that is used to describe the reactions between acids and bases. The theory is another way for one to look at the reaction between H+ and OH- ions or the reaction between acids and bases. This theory is the exact same idea as the Brønsted-Lowry Theory because it describes the reaction between acids and bases, however this theory describes the movement of the electron pairs in the reaction rather than the protons. This theory was another break through in the science world because scientists were able to understand another level of the reaction between acids and bases, they now understood the movement of the electron pairs.
Once the Brønsted-Lowry Theory was created, which brought a description of the movement of protons in an acid base reaction, scientists wondered what happened to the electrons during the reaction. The Lewis Theory, created by Gilbert N. Lewis, is another theory that is used to describe the reactions between acids and bases. The theory is another way for one to look at the reaction between H+ and OH- ions or the reaction between acids and bases. This theory is the exact same idea as the Brønsted-Lowry Theory because it describes the reaction between acids and bases, however this theory describes the movement of the electron pairs in the reaction rather than the protons. This theory was another break through in the science world because scientists were able to understand another level of the reaction between acids and bases, they now understood the movement of the electron pairs.
- All acids are electrons are electron pair receivers, meaning that during a reaction with a base they receive the base's electron pairs.
- All bases are electron pair donors, meaning that during a reaction with an acid they donate their electron pairs to the acid.
- Note that the definitions of acids and bases are exactly opposite from the definitions of the Brønsted-Lowry Theory because the acids are the receivers and the bases are the donors.