Strength of Acids and Bases
The strength of an acid is determined through its dissociation. The more that an acid dissociates in water, the more H+ ions are created, As a result. if an acid only partially dissociates, the solution will not create as many H+ ions. The strength of an acid is determined by the completion of the dissociation, which in turn also means that the strength of an acid is shown through the quantity of H+ ions in the chemical.
- Dissociation:
- Full dissociation= Hx --> H+ + x- One can tell that the dissociation is full because the arrow is used to show what the reaction yields.
- Example: HCl--> H2+ Cl This reaction shows a partial dissociation because the equilibrium sign is not used and the proper arrow is used.
- Partial dissociation= Hx ⇋ H+ + x- One can tell the this is partial dissociation because the equilibrium arrow is used to show what the reaction yields.
- Example: HCl⇋ H2+ Cl This reaction shows a partial dissociation because the equilibrium sign is used.
- Full dissociation= Hx --> H+ + x- One can tell that the dissociation is full because the arrow is used to show what the reaction yields.
- Dissociation:
- Full dissociation= MOH --> M+ + OH- One can tell that the dissociation is full because the arrow is used to show what the reaction yields.
- Example: NaOH --> Na + OH This reaction shows a partial dissociation because the equilibrium sign is not used and now the proper arrow is used to show the dissociation.
- Partial dissociation= MOH ⇋ M+ + OH- One can tell the this is partial dissociation because the equilibrium arrow is used to show what the reaction yields.
- Example: NaOH⇋ Na+ OH This reaction shows a partial dissociation because the equilibrium sign is used.
- Full dissociation= MOH --> M+ + OH- One can tell that the dissociation is full because the arrow is used to show what the reaction yields.
- Strengths of Acids:
- A strong acid will have a pH of 0-3. When an acid has a pH of 0-3 this means that the solution has completely dissociated and thus contains many H+ ions.
- H2SO4 is a stronger acid than HCl because it contains more H+ ions.
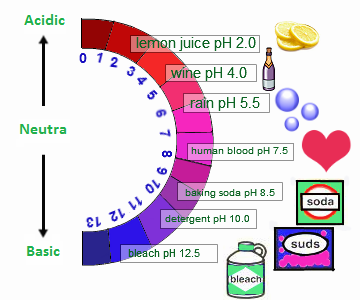
- Lemon Juice is a stronger acid than orange juice because lemon juice has a pH of 2 and orange juice has a pH of 4. Because lemon juice has a smaller pH, it means that the solution is more a stronger acid.
- A weak acid will have a pH of a 4-6. When an acid has a pH of 4-6 this means that the solution has not completely dissociated and as a result will not have as many H+ ions.
- HCl is a weaker acid than H2SO4 because it contains fewer H+ ions.
- Orange juice is a weaker acid than vinegar because orange juice has a pH of 4 and vinegar has a pH of 2. Because of orange juice's larger pH, it is a weaker acid than vinegar.
- A strong acid will have a pH of 0-3. When an acid has a pH of 0-3 this means that the solution has completely dissociated and thus contains many H+ ions.
- Strengths of Bases:
- A strong base will have a pH of 11-14. When a base has a pH of 11-14 this means that the solution has completely dissociated and thus contains many OH- ions.
- Na(OH)2 is a stronger base than NaOH because it contains fewer OH- ions.
- Bleach is a stronger base than baking soda because bleach has a pH of 11 and baking soda has a pH of 8. Because of bleach's larger pH, the solution is a stronger.
- A weak base will have a pH of 8-10. When a base has a pH of 8-10 this means that the solution has not completely dissociated and thus will contain fewer OH- ions.
- NaOH is a weaker base than Na(OH)2 because it contains fewer OH ions.
- Detergent is a weaker base than ammonia because detergent has a pH of 10 and ammonia has a pH of 11. Because of detergent's smaller pH, it is weaker.
- A strong base will have a pH of 11-14. When a base has a pH of 11-14 this means that the solution has completely dissociated and thus contains many OH- ions.