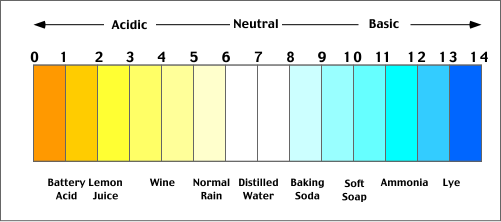
The pH Scale
Acids and bases are describing words, which are used to distinguish chemicals and their specific traits that differentiate them from another chemical. As explained previously in the journey, acids and bases have distinguished traits. The pH scale is used to classify the acids and bases and how weak or strong the specific chemical is. In other words, the scale measures how acidic or basic a specific substance is. The scale ranges from 0-14. An acid will have a pH of 0-6.9 where a pH of 0 is an extremely strong acid and a pH of 6 is a weak acid. A base will have a pH of 7.1-14 where a pH of 8 is a weak base and a pH of 14 is a strong base. A pH of 7 is neutral. The pH scale is logarithmic and as a result, each pH value under 7 is 10 times more acidic than the lower number. For example, a pH of 3 is 10 times more acidic than a pH of 4 and 100 times more acidic than a pH of 5. The same goes for pHs above 7, when increasing the pH, the higher pH will always be 10 times more basic than the previous pH. For example, a pH of 11 is 10 times more basic than a pH of 10 and is 100 times more basic than a pH of 9.