pH Indicators
Indicators (see photos at the bottom for clarification):
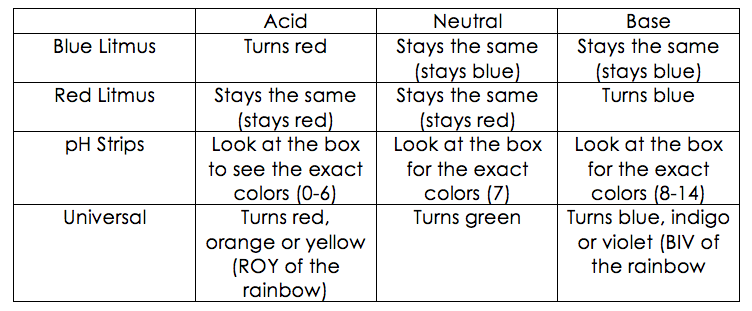
- Red litmus paper= a piece of paper that is coated with litmus that is red. When the paper is placed into an acid, the paper will remain red. When the paper is placed into a neutral, the paper will also remain red. However, when the paper is placed in a base, the paper will change blue.
- Blue litmus paper= a piece of paper that is coated with litmus that is blue. When the paper is placed into a base, the paper will remain blue. When the paper is placed into a neutral substance, the paper will also remain blue. However, when the paper is placed in an acid, the paper will change red.
- pH strips= a strip of paper that has shows different colors that coordinate to the box that the papers come in. When the strip is placed in a chemical, the colored bodes on the top will change colors. From there a scientist will use the key on the back to distinguish the pH of the chemical.
- Universal indicator= a chemical that when dropped into a substance will change colors according to the pH. One will be able to distinguish the pH of the liquid by seeing what color the substance changes into. If the universal indicator is dropped into an acid, then the chemical will change to either red, orange or yellow. The closer the color of the chemical is to deep red, the stronger the acid, meaning that the closer the acid is to yellow, the weaker the acid is. If the indicator is inserted into a neutral, then the substance will turn green. If the indicator is added to a base, then the color will turn blue, indigo or violet. The closer the color is to violet, the stronger the base, meaning that the closer the color is to blue, the weaker the base is.
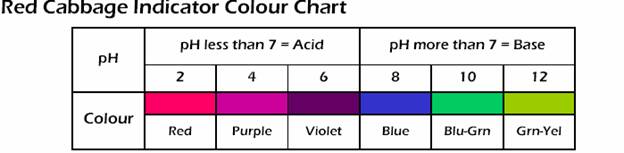
- Red Cabbage Indicator= This is another liquid indicator that is placed into a solution and the result is the changing of colors of the solution. The indicator is made by chopping up red cabbage into small pieces and then boiling it until the color translates into the water and for around 10 minutes. Next it is important to strain the cut up pieces of cabbage out of the liquid. Then when one pours the purple solution into the substance that is being tested, the liquid will change colors according to its pH. All acids range from the strong ones turning dark pink, the middle strength turing purple and the weak turning violet. Then for bases, the strong ones turn light green, the middle strength ones turn dark green and the weak turn dark blue. Neutral is light bluye.
Uses of the Indicators:
- Red litmus paper= they are best used when one simply wants to know if a liquid is an acid or a base, not the strength of the substance. However, when one uses red litmus paper, if the paper stays red, then one will not know that substance is either acidic or neutral, since the paper will stay red for both of circumstances. In that event, one can do a further test and use the blue litmus paper. If the paper turns red then the substance is an acid. If the paper stays blue then the substance is neutral.
- Blue litmus paper= they are also best used when one simply wants to know if a liquid is an acid or a base, not the strength of the substance. However, when one uses blue litmus paper, if the paper stays blue, then one will not know that substance is either basic or neutral, since the paper will stay blue when placed in both of those substances. In that event, one can do a further test and use the red litmus paper. If the paper turns blue then the substance is an base. If the paper stays red then the substance is neutral.
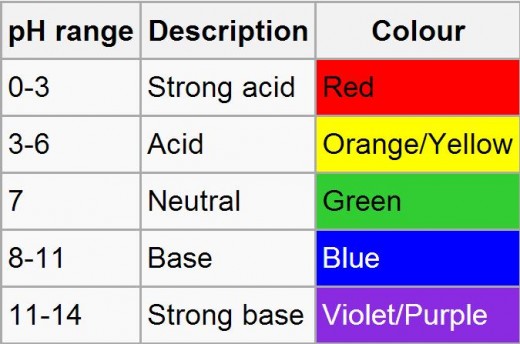
- pH strips= they are best used when one is interested in finding the exact pH of a chemical or liquid. When the pH strip is placed into the liquid, the end with the colored squares will change colors. Once that is done, one can compare the colors of the pH strip with the key on the back, which will allow the scientist to know what the exact pH of the liquid is.
- Universal Indicator= the indicator is best use when one has a lot of liquid and one wants to know the pH of the liquid but does not want to put his or her hand close enough to the liquid to place a strip into it. In this case, the universal indicator can be used when a large amount of liquid is being questioned. One can pour a small amount of the liquid and test the specimen separately. Once the specimen is separated, one can drop three drops of the indicator and view the change in the color, which will indicate the pH.
- Red Cabbage Indicator= the indicator is best used for the same circumstances as the universal indicator as they are both liquid chemicals that will change the color of a solution in order to present the pH to the scientist. However, this indicator is especially useful for someone who is not a scientist and does not have any indicators handy at home. If someone is intellectually curious and wants to know the pH of a liquid when he or she is at home, one is easily able to make this indicator with household items, this makes the red cabbage indicator unique because the rest must be purchased while this one is homemade.